Evaporation and the Water Cycle Completed
Evaporation is the process that changes liquid water to gaseous water (water vapor). Water moves from the Earth’s surface to the atmosphere via evaporation.
• Water Science School HOME • The Water Cycle •
Water cycle components » Atmosphere · Condensation · Evaporation · Evapotranspiration · Freshwater lakes and rivers · Groundwater flow · Groundwater storage · Ice and snow · Infiltration · Oceans · Precipitation · Snowmelt · Springs · Streamflow · Sublimation · Surface runoff

Credit: Alan Cressler, USGS
What is evaporation and why does it occur?
Evaporation is the process that changes liquid water to gaseous water (water vapor). Water moves from the Earth’s surface to the atmosphere via evaporation.
Evaporation occurs when energy (heat) forces the bonds that hold water molecules together to break. When you’re boiling water on the stove, you’re adding heat to liquid water. This added heat breaks the bonds, causing the water to shift from its liquid state to its gaseous state (water vapor), which we know as steam.
Water easily evaporates at its boiling point (212° F, 100° C) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the water.
The opposite of evaporation is condensation. Condensation is the process of water vapor turning back into liquid water. Condensation occurs when saturated air is cooled, such as on the outside of a glass of ice water.

Evaporation drives the water cycle
Most of the moisture in the atmosphere (about 90%) came from water evaporating from oceans, seas, lakes, and rivers. (And because over 70% of Earth’s surface is covered by oceans, they contribute a lot to the overall volume of water evaporating into the atmosphere.) The rest of the moisture in the atmosphere came from plant transpiration and (a very small amount) from sublimation.
On a global scale, the amount of water evaporating is about the same as the amount of water delivered to the Earth as precipitation.
This does vary geographically, though. Evaporation is more prevalent over the oceans than precipitation, while over the land, precipitation routinely exceeds evaporation. Most of the water that evaporates from the oceans falls back into the oceans as precipitation. Only about 10 percent of the water evaporated from the oceans is transported over land and falls as precipitation. Once evaporated, a water molecule spends about 10 days in the air. The process of evaporation is so great that without precipitation runoff, and groundwater discharge from aquifers, oceans would become nearly empty.

People make use of evaporation
One way to produce table salt is to evaporate saline water in evaporation ponds, a technique used by people for thousands of years.
Seawater contains other valuable minerals that are easily obtained due to evaporation. Water from the Dead Sea is ideal for the extraction of not only table salt, but also magnesium, potash, and bromine. The Dead Sea is actually a lake, located in the Middle East within a closed watershed and without any means of outflow. This closed basin system is abnormal for most lakes. Water primarily leaves the lake by evaporating, resulting in upwards of 1,300 - 1,600 millimeters of evaporated water per year in this desert area! The result is that the waters of the Dead Sea have the highest salinity and density of any sea in the world, too high to support life.
(Source: Overview of Middle East Water Resources, Middle East Water Data Banks Project).
Evaporative cooling: Cheap air conditioning!
Because energy is required to break the bonds holding water molecules together, evaporation removes heat from the environment, leading to a net cooling. Think of when a physician wipes an alcohol pad on your arm before giving you an injection – your arm feels cold. That’s because the alcohol rapidly evaporates from the surface of your skin, and with it some heat from the surface of your arm.
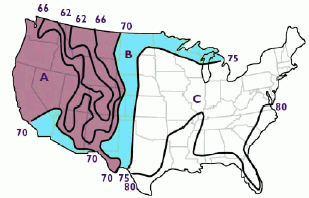
In climates where the humidity is low and the temperatures are hot, an evaporative cooler can lower the air temperature by 20 degrees F., while it increases humidity. As this map shows, evaporative coolers work best in the dry areas of the United States (red areas marked A) and can work somewhat in the blue areas marked B. But in section C, in the humid eastern U.S., normal air conditioners must be used.
Evaporative coolers are really quite simple devices, at least compared to air conditioners, because they pull in the dry, hot outdoor air and pass it through an evaporative pad that is kept wet by a supply of water. In a home device, a fan draws the air through the pad causing the water in the pad to evaporate, resulting in cooler air which is then pumped through the house. Much less energy is used as compared to an air conditioner.
Credit: California Energy Commission
Below are other science topics associated with the water cycle.
Precipitation and the Water Cycle
Streamflow and the Water Cycle
Snowmelt Runoff and the Water Cycle
The Atmosphere and the Water Cycle
Condensation and the Water Cycle
Infiltration and the Water Cycle
Surface Runoff and the Water Cycle
Springs and the Water Cycle
Sublimation and the Water Cycle
Ice, Snow, and Glaciers and the Water Cycle
Groundwater Flow and the Water Cycle
Groundwater Storage and the Water Cycle
Evaporation is the process that changes liquid water to gaseous water (water vapor). Water moves from the Earth’s surface to the atmosphere via evaporation.
• Water Science School HOME • The Water Cycle •
Water cycle components » Atmosphere · Condensation · Evaporation · Evapotranspiration · Freshwater lakes and rivers · Groundwater flow · Groundwater storage · Ice and snow · Infiltration · Oceans · Precipitation · Snowmelt · Springs · Streamflow · Sublimation · Surface runoff

Credit: Alan Cressler, USGS
What is evaporation and why does it occur?
Evaporation is the process that changes liquid water to gaseous water (water vapor). Water moves from the Earth’s surface to the atmosphere via evaporation.
Evaporation occurs when energy (heat) forces the bonds that hold water molecules together to break. When you’re boiling water on the stove, you’re adding heat to liquid water. This added heat breaks the bonds, causing the water to shift from its liquid state to its gaseous state (water vapor), which we know as steam.
Water easily evaporates at its boiling point (212° F, 100° C) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the water.
The opposite of evaporation is condensation. Condensation is the process of water vapor turning back into liquid water. Condensation occurs when saturated air is cooled, such as on the outside of a glass of ice water.

Evaporation drives the water cycle
Most of the moisture in the atmosphere (about 90%) came from water evaporating from oceans, seas, lakes, and rivers. (And because over 70% of Earth’s surface is covered by oceans, they contribute a lot to the overall volume of water evaporating into the atmosphere.) The rest of the moisture in the atmosphere came from plant transpiration and (a very small amount) from sublimation.
On a global scale, the amount of water evaporating is about the same as the amount of water delivered to the Earth as precipitation.
This does vary geographically, though. Evaporation is more prevalent over the oceans than precipitation, while over the land, precipitation routinely exceeds evaporation. Most of the water that evaporates from the oceans falls back into the oceans as precipitation. Only about 10 percent of the water evaporated from the oceans is transported over land and falls as precipitation. Once evaporated, a water molecule spends about 10 days in the air. The process of evaporation is so great that without precipitation runoff, and groundwater discharge from aquifers, oceans would become nearly empty.

People make use of evaporation
One way to produce table salt is to evaporate saline water in evaporation ponds, a technique used by people for thousands of years.
Seawater contains other valuable minerals that are easily obtained due to evaporation. Water from the Dead Sea is ideal for the extraction of not only table salt, but also magnesium, potash, and bromine. The Dead Sea is actually a lake, located in the Middle East within a closed watershed and without any means of outflow. This closed basin system is abnormal for most lakes. Water primarily leaves the lake by evaporating, resulting in upwards of 1,300 - 1,600 millimeters of evaporated water per year in this desert area! The result is that the waters of the Dead Sea have the highest salinity and density of any sea in the world, too high to support life.
(Source: Overview of Middle East Water Resources, Middle East Water Data Banks Project).
Evaporative cooling: Cheap air conditioning!
Because energy is required to break the bonds holding water molecules together, evaporation removes heat from the environment, leading to a net cooling. Think of when a physician wipes an alcohol pad on your arm before giving you an injection – your arm feels cold. That’s because the alcohol rapidly evaporates from the surface of your skin, and with it some heat from the surface of your arm.
In climates where the humidity is low and the temperatures are hot, an evaporative cooler can lower the air temperature by 20 degrees F., while it increases humidity. As this map shows, evaporative coolers work best in the dry areas of the United States (red areas marked A) and can work somewhat in the blue areas marked B. But in section C, in the humid eastern U.S., normal air conditioners must be used.
Evaporative coolers are really quite simple devices, at least compared to air conditioners, because they pull in the dry, hot outdoor air and pass it through an evaporative pad that is kept wet by a supply of water. In a home device, a fan draws the air through the pad causing the water in the pad to evaporate, resulting in cooler air which is then pumped through the house. Much less energy is used as compared to an air conditioner.

Credit: California Energy Commission
Below are other science topics associated with the water cycle.













