Dissolved Oxygen and Water Completed
Dissolved Oxygen and Water
Dissolved oxygen (DO) is a measure of how much oxygen is dissolved in the water - the amount of oxygen available to living aquatic organisms. The amount of dissolved oxygen in a stream or lake can tell us a lot about its water quality.
• Water Science School HOME • Water Properties topics • Water Quality topics •

The USGS has been measuring water for decades. Some measurements, such as temperature, pH, and specific conductance are taken almost every time water is sampled and investigated, no matter where in the U.S. the water is being studied. Another common measurement often taken is dissolved oxygen (DO), which is a measure of how much oxygen is dissolved in the water - DO can tell us a lot about water quality.
Dissolved Oxygen and Water
Although water molecules contain an oxygen atom, this oxygen is not what is needed by aquatic organisms living in natural waters. A small amount of oxygen, up to about ten molecules of oxygen per million of water, is actually dissolved in water. Oxygen enters a stream mainly from the atmosphere and, in areas where groundwater discharge into streams is a large portion of streamflow, from groundwater discharge. This dissolved oxygen is breathed by fish and zooplankton and is needed by them to survive.
Dissolved oxygen and water quality

Rapidly moving water, such as in a mountain stream or large river, tends to contain a lot of dissolved oxygen, whereas stagnant water contains less. Bacteria in water can consume oxygen as organic matter decays. Thus, excess organic material in lakes and rivers can cause eutrophic conditions, which is an oxygen-deficient situation that can cause a water body to "die." Aquatic life can have a hard time in stagnant water that has a lot of rotting, organic material in it, especially in summer (the concentration of dissolved oxygen is inversely related to water temperature), when dissolved-oxygen levels are at a seasonal low. Water near the surface of the lake– the epilimnion– is too warm for them, while water near the bottom–the hypolimnion– has too little oxygen. Conditions may become especially serious during a period of hot, calm weather, resulting in the loss of many fish. You may have heard about summertime fish kills in local lakes that likely result from this problem.
(Source: A Citizen's Guide to Understanding and Monitoring Lakes and Streams)
Dissolved oxygen, temperature, and aquatic life

As the chart shows, the concentration of dissolved oxygen in surface water is affected by temperature and has both a seasonal and a daily cycle. Cold water can hold more dissolved oxygen than warm water. In winter and early spring, when the water temperature is low, the dissolved oxygen concentration is high. In summer and fall, when the water temperature is high, the dissolved-oxygen concentration is often lower.
Dissolved oxygen in surface water is used by all forms of aquatic life; therefore, this constituent typically is measured to assess the "health" of lakes and streams. Oxygen enters a stream from the atmosphere and from groundwater discharge. The contribution of oxygen from groundwater discharge is significant, however, only in areas where groundwater is a large component of streamflow, such as in areas of glacial deposits. Photosynthesis is the primary process affecting the dissolved-oxygen/temperature relation; water clarity and strength and duration of sunlight, in turn, affect the rate of photosynthesis.
Hypoxia and "Dead zones"
You may have heard about a Gulf of Mexico "dead zone" in areas of the Gulf south of Louisiana, where the Mississippi and Atchafalaya Rivers discharge. A dead zone forms seasonally in the northern Gulf of Mexico when subsurface waters become depleted in dissolved oxygen and cannot support most life. The zone forms west of the Mississippi Delta over the continental shelf off Louisiana and sometimes extends off Texas. The oxygen depletion begins in late spring, increases in summer, and ends in the fall.
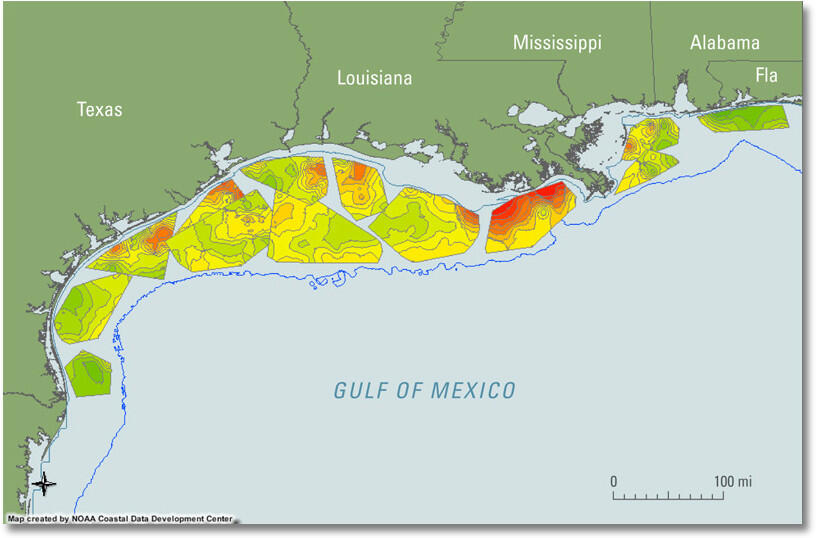
The formation of oxygen-depleted subsurface waters has been associated with nutrient-rich (nitrogen and phosphorus) discharge from the Mississippi and Atchafalaya Rivers. Bio-available nutrients in the discharge can stimulate algal blooms, which die and are eaten by bacteria, depleting the oxygen in the subsurface water. The oxygen content of surface waters of normal salinity in the summer is typically more than 8 milligrams per liter (8 mg/L); when oxygen concentrations are less than 2 mg/L, the water is defined as hypoxic (CENR, 2000). The hypoxia kills many organisms that cannot escape, and thus the hypoxic zone is informally known as the “dead zone.”
The hypoxic zone in the northern Gulf of Mexico is in the center of a productive and valuable fishery. The increased frequency and expansion of hypoxic zones have become an important economic and environmental issue to commercial and recreational users of the fishery.
Measuring dissolved oxygen

Field and lab meters to measure dissolved oxygen have been around for a long time. As this picture shows, modern meters are small and highly electronic. They still use a probe, which is located at the end of the cable. Dissolved oxygen is dependent on temperature (an inverse relation), so the meter must be calibrated properly before each use.
Do you want to test your local water quality?
Water test kits are available from World Water Monitoring Challenge (WWMC), an international education and outreach program that builds public awareness and involvement in protecting water resources around the world. Teachers and water-science enthusiasts: Do you want to be able to perform basic water-quality tests on local waters? WWMC offers inexpensive test kits so you can perform your own tests for temperature, pH, turbidity, and dissolved oxygen.
Do you think you know a lot about water properties?
Take our interactive water-properties true/false quiz and test your water knowledge.

Want to know more about dissolved oxygen and water? Follow me to the Nutrients and Eutrophication website!
Learn more about dissolved oxygen and related water topics.
Water Properties Information by Topic
pH and Water
Groundwater Flow and the Water Cycle
Temperature and Water
Conductivity (Electrical Conductance) and Water
Turbidity and Water
Harmful Algal Blooms (HABs)
Harmful Algae Blooms (HABs)
Transport and Fate of Nutrients
Below are multimedia items associated with dissolved oxygen and related water topics.
Below are publications associated with dissolved oxygen and water.
Chapter A6. Section 6.2. Dissolved oxygen
Gulf of Mexico dead zone —The last 150 years
- Overview
Dissolved oxygen (DO) is a measure of how much oxygen is dissolved in the water - the amount of oxygen available to living aquatic organisms. The amount of dissolved oxygen in a stream or lake can tell us a lot about its water quality.
• Water Science School HOME • Water Properties topics • Water Quality topics •
 Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsUSGS scientist is measuring various water-quality conditions in Holes Creek at Huffman Park in Kettering, Ohio. The USGS has been measuring water for decades. Some measurements, such as temperature, pH, and specific conductance are taken almost every time water is sampled and investigated, no matter where in the U.S. the water is being studied. Another common measurement often taken is dissolved oxygen (DO), which is a measure of how much oxygen is dissolved in the water - DO can tell us a lot about water quality.
Dissolved Oxygen and Water
Although water molecules contain an oxygen atom, this oxygen is not what is needed by aquatic organisms living in natural waters. A small amount of oxygen, up to about ten molecules of oxygen per million of water, is actually dissolved in water. Oxygen enters a stream mainly from the atmosphere and, in areas where groundwater discharge into streams is a large portion of streamflow, from groundwater discharge. This dissolved oxygen is breathed by fish and zooplankton and is needed by them to survive.
Dissolved oxygen and water quality
 Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsA eutrophic lake where dissolved-oxygen concentrations are low. Algal blooms can occur under such conditions. Rapidly moving water, such as in a mountain stream or large river, tends to contain a lot of dissolved oxygen, whereas stagnant water contains less. Bacteria in water can consume oxygen as organic matter decays. Thus, excess organic material in lakes and rivers can cause eutrophic conditions, which is an oxygen-deficient situation that can cause a water body to "die." Aquatic life can have a hard time in stagnant water that has a lot of rotting, organic material in it, especially in summer (the concentration of dissolved oxygen is inversely related to water temperature), when dissolved-oxygen levels are at a seasonal low. Water near the surface of the lake– the epilimnion– is too warm for them, while water near the bottom–the hypolimnion– has too little oxygen. Conditions may become especially serious during a period of hot, calm weather, resulting in the loss of many fish. You may have heard about summertime fish kills in local lakes that likely result from this problem.
(Source: A Citizen's Guide to Understanding and Monitoring Lakes and Streams)
Dissolved oxygen, temperature, and aquatic life
 Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsWater temperture affects dissolved-oxygen concentrations in a river or water body. As the chart shows, the concentration of dissolved oxygen in surface water is affected by temperature and has both a seasonal and a daily cycle. Cold water can hold more dissolved oxygen than warm water. In winter and early spring, when the water temperature is low, the dissolved oxygen concentration is high. In summer and fall, when the water temperature is high, the dissolved-oxygen concentration is often lower.
Dissolved oxygen in surface water is used by all forms of aquatic life; therefore, this constituent typically is measured to assess the "health" of lakes and streams. Oxygen enters a stream from the atmosphere and from groundwater discharge. The contribution of oxygen from groundwater discharge is significant, however, only in areas where groundwater is a large component of streamflow, such as in areas of glacial deposits. Photosynthesis is the primary process affecting the dissolved-oxygen/temperature relation; water clarity and strength and duration of sunlight, in turn, affect the rate of photosynthesis.
Hypoxia and "Dead zones"
You may have heard about a Gulf of Mexico "dead zone" in areas of the Gulf south of Louisiana, where the Mississippi and Atchafalaya Rivers discharge. A dead zone forms seasonally in the northern Gulf of Mexico when subsurface waters become depleted in dissolved oxygen and cannot support most life. The zone forms west of the Mississippi Delta over the continental shelf off Louisiana and sometimes extends off Texas. The oxygen depletion begins in late spring, increases in summer, and ends in the fall.
 Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsDissolved oxygen in bottom waters, measured from June 8 through July 17, 2009, during the annual summer Gulf of Mexico Southeast Area Monitoring and Assessment Program (SEAMAP) cruise in the northern Gulf of Mexico. Orange and red colors indicate lower dissolved oxygen concentrations. The formation of oxygen-depleted subsurface waters has been associated with nutrient-rich (nitrogen and phosphorus) discharge from the Mississippi and Atchafalaya Rivers. Bio-available nutrients in the discharge can stimulate algal blooms, which die and are eaten by bacteria, depleting the oxygen in the subsurface water. The oxygen content of surface waters of normal salinity in the summer is typically more than 8 milligrams per liter (8 mg/L); when oxygen concentrations are less than 2 mg/L, the water is defined as hypoxic (CENR, 2000). The hypoxia kills many organisms that cannot escape, and thus the hypoxic zone is informally known as the “dead zone.”
The hypoxic zone in the northern Gulf of Mexico is in the center of a productive and valuable fishery. The increased frequency and expansion of hypoxic zones have become an important economic and environmental issue to commercial and recreational users of the fishery.
Measuring dissolved oxygen
 Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsMulti-parameter monitor used to record water-quality measurements. Field and lab meters to measure dissolved oxygen have been around for a long time. As this picture shows, modern meters are small and highly electronic. They still use a probe, which is located at the end of the cable. Dissolved oxygen is dependent on temperature (an inverse relation), so the meter must be calibrated properly before each use.
Do you want to test your local water quality?
Water test kits are available from World Water Monitoring Challenge (WWMC), an international education and outreach program that builds public awareness and involvement in protecting water resources around the world. Teachers and water-science enthusiasts: Do you want to be able to perform basic water-quality tests on local waters? WWMC offers inexpensive test kits so you can perform your own tests for temperature, pH, turbidity, and dissolved oxygen.
 Sources/Usage: Some content may have restrictions. View Media Details
Sources/Usage: Some content may have restrictions. View Media DetailsDo you think you know a lot about water properties?
Take our interactive water-properties true/false quiz and test your water knowledge. Sources/Usage: Public Domain. View Media Details
Sources/Usage: Public Domain. View Media DetailsWant to know more about dissolved oxygen and water? Follow me to the Nutrients and Eutrophication website!
- Science
Learn more about dissolved oxygen and related water topics.
Water Properties Information by Topic
Looking at water, you might think that it's the most simple thing around. Pure water is practically colorless, odorless, and tasteless. But it's not at all simple and plain and it is vital for all life on Earth. Where there is water there is life, and where water is scarce, life has to struggle or just "throw in the towel." Continue on to learn about dozens of water properties.pH and Water
pH is a measure of how acidic/basic water is. The range goes from 0 to 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base. The pH of water is a very important measurement concerning water quality.Groundwater Flow and the Water Cycle
Yes, water below your feet is moving all the time, but not like rivers flowing below ground. It's more like water in a sponge. Gravity and pressure move water downward and sideways underground through spaces between rocks. Eventually it emerges back to the land surface, into rivers, and into the oceans to keep the water cycle going.Temperature and Water
Water temperature plays an important role in almost all USGS water science. Water temperature exerts a major influence on biological activity and growth, has an effect on water chemistry, can influence water quantity measurements, and governs the kinds of organisms that live in water bodies.Conductivity (Electrical Conductance) and Water
Water and electricity don't mix, right? Well actually, pure water is an excellent insulator and does not conduct electricity. The thing is, you won't find any pure water in nature, so don't mix electricity and water. Our Water Science School page will give you all the details.Turbidity and Water
Lucky for us all, our drinking water is almost always clear (very low turbidity). Other water, such as the creek behind your house after a rainstorm, is likely to be highly turbid—brown with floating sediment. Turbidity is the clarity of water and it is an important factor in water quality.Harmful Algal Blooms (HABs)
The USGS collaborates with local, state, federal, tribal, university, and industry partners to conduct the science necessary to understand the causes and effects of toxic HABs and inform water management and public health decisions. USGS is characterizing the life cycle of HABs, their asociated toxins, and the genes responsible for cyanotoxin production. This work is enhancing the ability of Great...Harmful Algae Blooms (HABs)
Cyanobacterial harmful algal blooms (HABs) are increasingly a global concern because HABs pose a threat to human and aquatic ecosystem health and cause economic damages. Toxins produced by some species of cyanobacteria (called cyanotoxins) can cause acute and chronic illnesses in humans and pets. Aquatic ecosystem health also is affected by cyanotoxins, as well as low dissolved oxygen...Transport and Fate of Nutrients
Eutrophication, or excess nutrients in streams, is typically one of the top reasons that a stream is listed as impaired on the 303(d) list as part of the Clean Water Act. How nutrients, primarily nitrogen and phosphorus, are transported to streams and groundwater greatly affects the best management plan to keep them on fields and out of streams and groundwater. Likewise, environmental managers and... - Multimedia
Below are multimedia items associated with dissolved oxygen and related water topics.
No results found. - Publications
Below are publications associated with dissolved oxygen and water.
Chapter A6. Section 6.2. Dissolved oxygen
Accurate data for the concentration of dissolved oxygen in surface and ground waters are essential for documenting changes in environmental water resources that result from natural phenomena and human activities. Dissolved oxygen is necessary in aquatic systems for the survival and growth of many aquatic organisms and is used as an indicator of the health of surface-water bodies. This section of tAuthorsStewart A. Rounds, Franceska D. Wilde, George F. RitzGulf of Mexico dead zone —The last 150 years
'Gulf of Mexico Dead Zone-The Last 150 Years' discusses the dead zone that forms seasonally in the northern Gulf of Mexico when subsurface waters become depleted in dissolved oxygen and cannot support most life.AuthorsLisa Osterman, P.W. Swarzenski, R. Z. Poore









